 The electron configuration of an element with an atomic number greater than 18 cannot be properly determined according to the Bohr atomic model. Electrons can be arranged correctly through orbits from elements 1 to 18. Therefore, the order of the number of electrons in each shell of the cobalt(Co) atom is 2, 8, 15, 2. Therefore, the cobalt atom will have two electrons in the first shell, and eight in the 2nd orbit.Īccording to Bohr’s formula, the third orbit will have seventeen electrons but the third orbit of cobalt will have fifteen electrons and the remaining two electrons will be in the fourth orbit. That is, the number of electrons in cobalt is twenty-seven. The atomic number is the number of electrons in that element. Therefore, the maximum electron holding capacity in the first shell is two, the second shell is eight and the 3rd shell can have a maximum of eighteen electrons.Ĭobalt atom electron configuration (Bohr model) The maximum electrons holding capacity in N orbit is 2n 2 = 2 × 4 2 = 32. The maximum electrons holding capacity in M orbit is 2n 2 = 2 × 3 2 = 18. The maximum electron holding capacity in L orbit is 2n 2 = 2 × 2 2 = 8. The maximum electron holding capacity in K orbit is 2n 2 = 2 × 1 2 = 2. The electron holding capacity of each orbit is 2n 2. K is the name of the first orbit, L is the second, M is the third, and N is the name of the fourth orbit. These circular paths are called orbit(shell). The electrons of the atom revolve around the nucleus in a certain circular path. The complete idea of the orbit is given there. 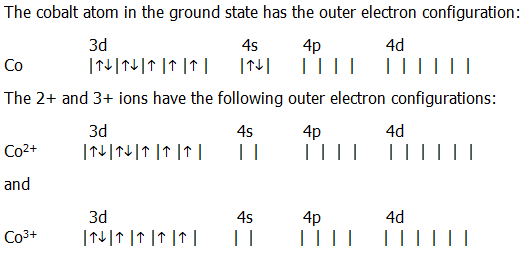
Scientist Niels Bohr was the first to give an idea of the atom’s orbit. Cobalt ion(Co2+, Co3+)electron configurationĬobalt atom electron configuration through orbit.How to write the orbital diagram for cobalt?.Electron configuration of cobalt through orbital.Cobalt atom electron configuration through orbit. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
